Hippocratic Medicine No. 5: The Heart of the Matter
It’s 7PM, and you are coming onto a busy 12-hour overnight shift. Your first patient is a 57-year-old African American female, Mrs. S., with a past medical history of hypertension. She is there with left sided chest pain intermittently present for the last 7-10 days that is not exertional, and not associated with any other symptoms. It lasts anywhere from minutes to an hour. She was evaluated for this pain last week, and a pulmonary embolism work-up was negative. She was discharged home with diagnosis of Left Pectoralis Muscle Strain. Her vital signs and exam are normal except for reproducible chest pain along the left rib margin. Her EKG demonstrates a normal sinus rhythm, and her first troponin is negative.
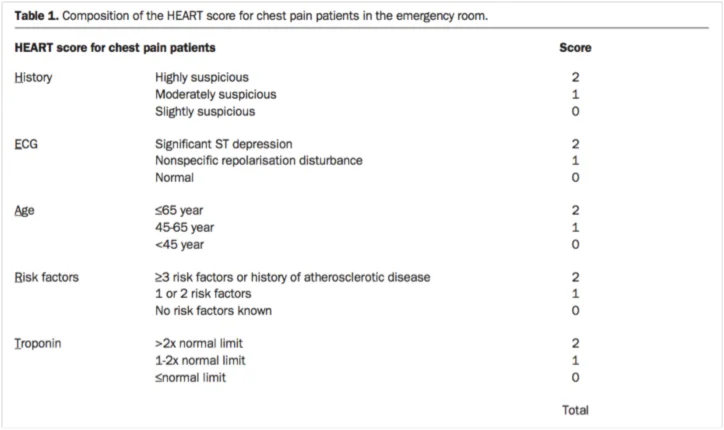
You decide to pursue a cardiac work-up with serial troponins and a cardiac stress echocardiogram. The patient is surprised because she has never been told she has high cholesterol, diabetes, or that she is otherwise at risk for atherosclerotic heart disease, but she agrees with this plan. While several risk stratification tools such as the Thrombosis in Myocardial Infarction (TIMI) risk score and Global Registry of Acute Coronary Events (GRACE) have been developed, they have not been sensitive enough to rule out Acute Coronary Syndrome (ACS) in ED patients [1, 2]. The HEART score (see table below) is a decision aid used to risk-stratify patients with a score that is interpreted on a range from low to high-risk for Major Adverse Cardiac Events (MACE). A cohort study by Mahler et al in the U.S. found a rate of MACE in 0.6% of 1070 patients presenting the ED with chest pain deemed to be low-risk by HEART score [2]. Mrs. S had a HEART score of 2, and would have been deemed low risk.
Mrs. S was amenable to the plan of further cardiac evaluation. However, if she was told she was low risk and given the option to follow up as an outpatient, would she have opted for this instead? Hess et al conducted a prospective, randomized study in which patients were given a pictograph and allowed to make this decision (see figure below). Compared to patients that received usual care (MD decision for ACS work up), those given the pictograph demonstrated better knowledge of their ACS risk, participated more in decision making, and opted less often to be admitted for further cardiac evaluation. Of note, there were no adverse cardiac events in either group [3].
Three days later, you notice that Mrs. S is still an inpatient. She had a positive stress test and subsequently underwent a cardiac catheterization that was negative for any significant coronary vessel disease. Post catheterization, Vascular Surgery was consulted because she had unfortunately developed a femoral artery pseudoaneurysm. Femoral artery vascular complications such as pseudoaneurysm and hematomas occur in up to 6% of cases, and other complications include dissection, thrombosis and infection [4,5,6].
The Coronary Artery Surgery Study (CASS) found that the pretest probability of coronary artery disease (CAD) varied between 5% and 89% based on age, sex, and chest pain characteristics [7]. Risk factors for CAD include chest pain consistent with angina, age, gender, history of diabetes or hyperlipidemia, and smoking. The current American College of Cardiology/American Heart Association guidelines for stress testing recommend assessment with an imaging modality (i.e. stress ECHO) for patients that are at intermediate risk for CAD. Any of the following factors suggests an intermediate risk: Prior history of CAD, ECG with ST-segment depression 0.05 to <0.10 mV and/or flat or inverted T waves <0.20 mV deep, diabetes mellitus, chronic kidney disease, and advanced age [8, 9, 10, 11].
With the rising costs of healthcare in the U.S., the American College of Physicians has called for more judicious use of screening and diagnostic tests. One principle they emphasize is that when the pretest probability of disease is low, the likelihood of a false positive test result is higher than a true positive test result [12]. Namely, cardiac stress testing in patients that are low risk for CAD is not recommended because the low pretest probability of this study in this population leads to subsequent higher likelihood for false positives and increased needless downstream interventions with their own rates of harm. Of note, the inverse is also true; those with a very high pretest probability are more likely to have a false negative result. Moreover, a retrospective study in 2013 by Foy et al demonstrated that out of 2662 patients without a history of CAD that had a cardiac catheterization, 866 were preceded by a stress imaging, with a pretest probability of 18% PPV for stress echocardiogram, and 27% for single-photon emission computed tomography. While this study is limited by the biases inherent to a retrospective study, it does suggest that too many stress tests are currently being ordered on low risk patients [13].
Mrs. S was low risk by her HEART score of 2 and had a low pretest probability for CAD based on the data from the CASS study, and was therefore at higher risk for a false positive result with cardiac stress testing. Furthermore, the downstream testing as a result of a positive stress test puts patients at risk for adverse events due to the invasive nature of cardiac catheterization and its complications. Mrs. S intuitively suspected that her risk for CAD was low, and as her physicians we should have easily and accurately made that assessment ourselves. As demonstrated by Hess et al, a better approach would have been to engage the patient in shared decision-making, which would have given her input into the management plan, and likely avoided this complication. While some physicians aim to reduce medico-legal liability by ruling out ACS with a stress test, our patients are exposed to over testing and even injury when this test is used indiscriminately. The cardiac stress test is to be used as a diagnostic tool in the correct clinical context, and a false positive result in a patient with a low pretest probability of disease may not only lead to complications, but also could have been determined without the test altogether.
Submitted by Aldo Andino, PGY-3
Peer Reviewed by Alicia Oberle, PGY-4
Faculty Reviewed by Douglas Char
Professor, Emergency Medicine, Washington University School of Medicine
References:
[1] Emerg Med J. 2014 Apr;31(4):281-5. doi: 10.1136/emermed-2012-201323. Epub 2013 Apr 10.
[2] Crit Pathw Cardiol. 2011 Sep;10(3):128-33.
[3] Circ Cardiovasc Qual Outcomes. 2012 May;5(3):251-9.
[4] Am J Cardiol. 1992;69(1):63.
[5] Am J Cardiol. 1993;72(13):47E.
[6] Circulation. 1993;88(4 Pt 1):1569.
[7] N Engl J Med. 1979;301:230-235.
[8] J Am Soc Echocardiogr. 2011 Mar;24(3):229-67. doi: 10.1016/j.echo.2010.12.008.
[9] J Am Coll Cardiol. 2002;40(8):1531.
[10] Circulation. 2009;119:e561-e587.
[11] J Am Soc Echocardiogr. 2011 Mar;24(3):229-67.
[12] Ann Intern Med. 2012 Jan 17;156(2):147-9.
[13] Am J Med Qual. 2014 Mar-Apr;29(2):153-9
The Hippocratic Medicine series is modeled after the Do No Harm project pioneered at the University of Colorado. The aim of this series is to raise awareness about the avoidance of avoidable care i.e. for how medical overuse has the potential to do patient harm. Because every test or intervention we do has the potential for not just benefit but also harm, we should seek that our patients do better because of the care we provide instead of despite it. The WUSM Hippocratic Medicine Series is supported by a grant provided by the Foundation for Barnes Jewish Hospital.